The physico-chemical basis of life
In 1944, the physicist
Erwin Schroedinger, winner of the Nobel
Prize in Physics in 1933 (together with Paul Dirac) published a short
volume called 'What is Life?' summarizing the emerging atomistic
foundation of the biological sciences in the 1930s and '40s. Schroedinger
proposed a correlation of the physical properties of DNA as an aperiodic
'crystal' with its function of storing the genetic information of every
living organism. A few years later in 1953, Francis Crick and James Watson
solved the high-resolution structure of DNA confirming Schroedinger's
theoretical considerations, while themselves proposing a mechanism of
DNA replication based on the structure of this molecule. A decade after
Watson and Crick, the syntax of DNA, the genetic code, had been determined,
and the biological, chemical, and physical pieces quickly fell into place
establishing a picture of molecular biology which matured into a scientific
fact establishing the foundation of the tremendously successful biotechnology.
|
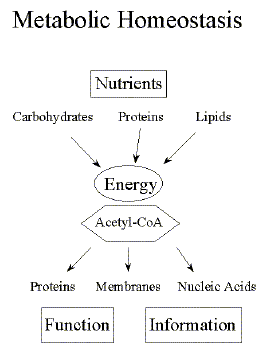
What is metabolic homeostasis? Fifty years after Schroedinger's publication, the biophysical and biochemical details of many cellular mechanisms are well characterized largely confirming Schroedinger's critical analysis of the limitations of the 'naive physicist's view' as applied to the life sciences. Contrary to the objects of physics, biological systems are complex in their composition, asymmetric in structure, and the flow of energy and chemical reactions never at equilibrium. Yet, organisms maintain a stable structure over long periods of time. This stability of large structures with the molecular components constantly replaced and regenerated is called homeostasis, meaning they maintain a status quo. Living organisms maintain a state of metabolic homeostasis which can be viewed as a steady-state throughput of energy to sustain body function, structure, and information. Both chemical energy and molecular building blocks are extracted from the nutrients in our diet and are used to form macromolecules like proteins, DNA, polysaccharides, and membranes. These macromolecular structures in turn control nutrient uptake, metabolism, and excretion. Metabolism is a dynamic process of energy conversion in cells of living organisms. In a most abstract representation, cellular metabolism is a spatial and temporal network of chemical reactions and molecular interactions close to, but never quite at chemical equilibrium. |
 |
Schroedinger's question
In 'What is Life?' Erwin Schroedinger asked for a physical and chemical accountability of the spatial and temporal processes occurring within the boundaries of a cell. As he made vividly clear, he deemed contemporary physics inappropriate to explain molecular mechanisms of a cell. Physical natural laws are exact only for large and very simple systems, like highly symmetric salt crystals. Furthermore, the most suitable physical theories to study cellular processes -- thermodynamics and quantum mechanics -- are statistical theories. They explain the behavior of large numbers of identical molecules, while they cannot describe the exact behavior of such individual molecules, but only give probabilities.
In 'What is Life?' Schroedinger outlined the naive physicist's view of life. Atoms and molecules are very small as compared to the size of an organism. The 'naive' conclusion from statistical physical laws is that for a complex system to be orderly, it better be large. This simply reflects the observation that addition or removal of a single unit from a system affects the overall system much more if there are few molecules (small N) than if their are millions of this unit around (large N). For the latter, we could hardly tell a difference when adding one to one billion. This phenomena can neatly be mathematically described. The random distribution away from the mean follows the square root of N and not N itself, making large systems predictable, while probabilities have to be assigned for small ones. This distinction between large numbers and small numbers is eminently important to understand biological systems, because they are small number systems rather than the convenient large number systems that physicists prefer. Already in 1944 it was clear that '..incredibly small groups of atoms, much too small to display exact statistical laws, do play a dominating role in the very orderly and lawful events within a living organism.'
Today we know that cellular processes are driven precisely by these discernible differences in molecular compositions. What statistical physics describes as fluctuation is a signaling process to cell biology. We are no more concerned about the size of the system, as we are about the size of the fluctuation. The size of random fluctuations can no longer be neglected, because in small systems they are significant with respect to the total size of the system. Single molecular events can trigger a cell division, fatty acid synthesis, or glucose degradation. Hormonal signals depend on the behavior of small systems, where fluctuations can push a system beyond a threshold level where a chemical reaction suddenly becomes spontaneous. Because biological systems are so extremely sensitive to small fluctuations, they can also react very quickly to changes in the environment.
Copyright © 2000-2009 Lukas K. Buehler